Introduction
Lower respiratory tract infections (LRTIs) cause millions of deaths globally each year and remain a major health and economic burden, especially in low- and middle-income countries. They are classified as community-acquired pneumonia (CAP) or nosocomial pneumonia (HAP/VAP). CAP is commonly caused by Streptococcus pneumoniae, while HAP/VAP often involve MRSA, Pseudomonas aeruginosa, and resistant Gram-negative bacteria. Proper classification and pathogen knowledge are essential for guiding empirical therapy, especially amid rising antimicrobial resistance. This review outlines current antibiotic options, summarizes these newly approved antibiotics and highlights emerging investigational therapies.
Current Guidelines
- Community-Acquired Pneumonia treatment is empirical and based on patient comorbidities, risk factors, and severity (using PSI).
- Healthy outpatients: amoxicillin, doxycycline, or macrolide (only if local resistance <25%).
- Outpatients with comorbidities: amoxicillin/clavulanate + macrolide/doxycycline, or fluoroquinolone monotherapy.
- Hospitalized non-severe CAP: β-lactam + macrolide, or respiratory fluoroquinolone.
- Severe CAP: β-lactam + macrolide preferred; fluoroquinolones now less recommended.
- Add MRSA or Pseudomonas coverage only if previously isolated or based on local risk factors.
- HAP/VAP therapy depends on MDR risk:
- Low-risk: ertapenem, ceftriaxone, cefotaxime, moxifloxacin, or levofloxacin.
- High-risk (not in shock): ICU monotherapy with broad antipseudomonal agent; add MRSA coverage if local MRSA >25%.
- Septic shock: dual antipseudomonal therapy + MRSA coverage; carbapenems preferred if ESBL common.
New Antibiotics for LRTIs
To address the growing challenge of antimicrobial resistance, several novel drugs with valuable therapeutic characteristics have been approved for the treatment of pneumonia.
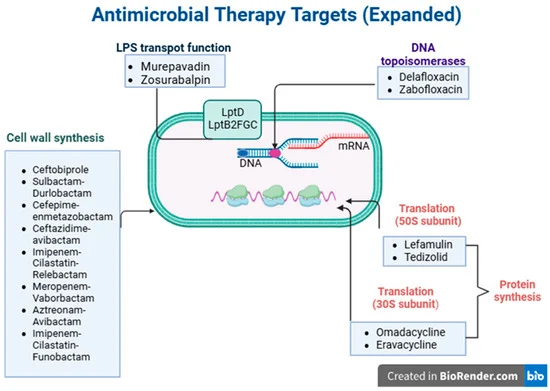
Figure 1: Antimicrobial therapy targets in the bacterial cell.
Antibiotic Agents for Community-Acquired Pneumonia
Lefamulin
Lefamulin is a new pleuromutilin antibiotic that blocks bacterial protein synthesis by binding to the 50S ribosome and has strong activity against typical and atypical CAP pathogens, including MDR S. pneumoniae and MRSA. Clinical trials (LEAP 1 & 2) showed it is non-inferior to moxifloxacin with similar cure rates and mainly mild gastrointestinal side effects. It penetrates lung tissue well, maintains effective ELF concentrations, and does not require dose adjustment in renal impairment, dialysis, cystic fibrosis, or most elderly patients. It may also have immune-modulating effects based on animal models, but human data are limited. Its main limitation is the very high cost compared to older antibiotics, though it is FDA and EMA approved for CAP treatment.
Omadacycline
Omadacycline is a next-generation tetracycline that overcomes common tetracycline resistance mechanisms and targets a wide range of CAP pathogens, including atypicals. In the OPTIC trial, it proved non-inferior to moxifloxacin with similar cure rates but fewer gastrointestinal side effects and no C. difficile cases. It achieves good concentrations in epithelial lining fluid and supports effective IV-to-oral and oral-only treatment, with no need for dose adjustment in renal or hepatic impairment, elderly patients, or most special populations. It has shown activity against Mycobacterium abscessus and severe Chlamydia psittaci pneumonia in real-world studies, and early evidence suggests possible anti-inflammatory effects. Although acquisition costs are higher, reduced hospital stay can offset expenses. FDA-approved for CABP and ABSSSI.
Delafloxacin
Delafloxacin is an anionic fluoroquinolone that inhibits both DNA gyrase and topoisomerase IV equally, making resistance less likely and providing broad coverage against Gram-positive (including MRSA), Gram-negative, and atypical pathogens. In phase III trials, it was non-inferior to moxifloxacin for CAP and showed better responses in patients with COPD or asthma. It demonstrates good lung penetration, does not prolong the QTc interval, and is generally well tolerated, with diarrhea and mild liver enzyme elevations being the most common adverse effects. Dose reduction is required only for the IV form in severe renal impairment, while the oral form does not need adjustment. It is approved in both the US and EU for CAP.
Ceftobiprole
Ceftobiprole is a fifth-generation cephalosporin approved for CAP and non-ventilator HAP, especially in frail patients with low–medium MDR risk who cannot tolerate anti-MRSA agents like vancomycin or linezolid. It offers broad coverage—including MRSA, VRSA, PRSP, Enterococcus faecalis, non-ESBL Enterobacterales, and P. aeruginosa—making it a useful carbapenem-sparing option, though it remains ineffective against ESBL-producers and carbapenemase-producing strains. Clinical trials show non-inferiority to ceftriaxone and ceftazidime/linezolid except in VAP, where outcomes are lower. Real-world data highlight good cure rates in elderly or immunocompromised patients, around 70–80% in ICU settings, with limited emergence of new MDR organisms. It maintains high MRSA/MSSA susceptibility (>95%), shows similar PK/PD in children and adults, is generally well tolerated, and is considered cost-neutral or cost-saving compared to standard regimens.
Antibiotic Agents for Nosocomial Pneumonia
Cefiderocol
Cefiderocol is a novel siderophore cephalosporin that uses a “Trojan horse” iron-uptake mechanism to enter Gram-negative bacteria, giving it strong activity against MDR and carbapenem-resistant pathogens like CRAB, CRE, P. aeruginosa, and S. maltophilia. It shows good lung penetration and is dosed at 2 g q8h via a 3-hour infusion, with adjustments based on kidney function. Clinical trials and real-world studies show non-inferiority to meropenem and better safety than colistin, though outcomes in A. baumannii infections have been variable. Guidelines recommend using cefiderocol cautiously and mainly in combination therapy for CRAB or MBL-producing organisms when other options are failing.
Ceftolozane/Tazobactam
Ceftolozane/tazobactam is a potent cephalosporin/β-lactamase inhibitor combination with strong activity against MDR and XDR Pseudomonas aeruginosa and ESBL-producing Enterobacterales. It achieves good lung penetration and is recommended at 3 g q8h for HAP/VAP. Clinical trials like ASPECT-NP confirm non-inferiority to meropenem, with strong real-world success in severe P. aeruginosa pneumonia and lower kidney toxicity compared with aminoglycosides or polymyxins. It is ineffective against carbapenemase-producers, so guidelines advise reserving it for difficult P. aeruginosa or as a carbapenem-sparing option.
Ceftazidime/Avibactam
Ceftazidime/avibactam combines a third-generation cephalosporin with a novel β-lactamase inhibitor effective against KPC, ESBL, AmpC, and some OXA enzymes, but not MBLs. It covers many resistant Enterobacterales and P. aeruginosa but lacks activity against A. baumannii and S. maltophilia. With good ELF penetration, it is dosed at 2.5 g q8h via 3-hour infusion. The REPROVE trial showed non-inferiority to meropenem in HAP/VAP with good tolerability. Real-world evidence supports its use as a key treatment for carbapenem-resistant Enterobacterales infections and as part of combination regimens when resistance patterns are uncertain.
Meropenem/Vaborbactam
Meropenem/vaborbactam combines meropenem with vaborbactam, a boron-based β-lactamase inhibitor that restores activity against KPC, ESBL, and AmpC-producing Enterobacterales, especially CRE. It has good lung penetration and is ineffective against A. baumannii, S. maltophilia, or P. aeruginosa. In the TANGO II trial, it showed higher cure rates and lower nephrotoxicity compared to best available therapy. Real-world data show early use leads to high success (≈80%) and low mortality, including in CAZ-AVI–resistant KPC infections. It is EMA-approved for HAP/VAP but not FDA-approved. IDSA recommends MVB mainly for carbapenem-resistant infections.
Imipenem/Cilastatin/Relebactam
Imipenem/cilastatin/relebactam restores imipenem activity against ESBL, AmpC, KPC-producing Enterobacterales and drug-resistant P. aeruginosa, but not A. baumannii or S. maltophilia. Both drugs achieve good ELF penetration. RESTORE-IMI 1 and 2 trials show non-inferiority to standard therapy, better outcomes in HAP/VAP, and significantly less nephrotoxicity compared with colistin-based regimens. Real-world data confirm 60–70% success, especially in DTR P. aeruginosa. FDA and EMA have approved it for HAP/VAP, and cost-effectiveness analyses favor IMI/REL despite higher drug costs.
Sulbactam/Durlobactam
Sulbactam/durlobactam is a targeted combination active against Acinetobacter baumannii complex, including carbapenem-resistant strains. Durlobactam protects sulbactam from class A, C, and D β-lactamase degradation, providing strong in vitro activity. The ATTACK trial showed non-inferiority to colistin with significantly lower nephrotoxicity and good ELF penetration. It works well in combination regimens without antagonism, including with imipenem or cefiderocol. FDA approved it in 2023 for HABP/VABP caused by A. baumannii, though high cost may limit access in low-resource regions.
Cefepime/Enmetazobactam
Cefepime/enmetazobactam is a newly approved combination for HAP/VAP that targets ESBL-producing Enterobacterales and shows activity against organisms with OXA-48 and AmpC enzymes. Pharmacokinetic studies confirm good ELF penetration and effective lung concentrations. Surveillance data show >97% susceptibility among ESBL-producing Enterobacterales, though it is ineffective against KPC producers. Its broad activity and safety profile make it a promising option for nosocomial pneumonia due to ESBL and AmpC organisms.
Aztreonam/Avibactam
Aztreonam/avibactam (ATM/AVI) combines a monobactam stable to MBLs with a β-lactamase inhibitor that protects it from ESBLs, AmpC, KPC, and OXA-48 enzymes, giving it strong activity against MBL-producing Enterobacterales and S. maltophilia. Large surveillance studies show it inhibits >99% of Enterobacterales, including CRE and MDR strains, and restores aztreonam susceptibility in most S. maltophilia isolates. Recommended dosing uses a loading dose followed by prolonged 3-hour infusions q6h, with renal adjustments. The REVISIT phase 3 trial showed clinical cure and microbiologic success comparable to meropenem, with lower mortality in HAP/VAP and good tolerability. Early data from the ASSEMBLE study also showed benefit in severe MBL infections. EMA approved ATM/AVI in April 2024 for cIAI, cUTI, and HAP/VAP when limited options exist.
Antibiotics Awaiting Approval
Tedizolid
Tedizolid is an oxazolidinone with strong activity against Gram-positive bacteria, including MRSA and some linezolid-resistant strains. It has excellent lung penetration and fewer GI and hematologic side effects than linezolid. A phase III study showed it was non-inferior for mortality in ventilated HAP/VAP but did not meet clinical cure non-inferiority. Thrombocytopenia can occur and requires monitoring. It is not FDA/EMA-approved for pneumonia.
Eravacycline
Eravacycline is a fluorocycline active against many MDR pathogens (MRSA, VRE, CRE, A. baumannii) but not P. aeruginosa. It has good lung penetration. Clinical data in A. baumannii pneumonia are mixed, partly due to sicker patients in studies. Resistance can develop through efflux pump overexpression. Adverse effects include GI symptoms and infusion-site reactions. More evidence is needed for pneumonia use.
Murepavadin
Murepavadin is a first-in-class antibiotic targeting P. aeruginosa outer membrane protein LptD. IV development stopped due to high nephrotoxicity in phase III trials. Research is now focused on an inhaled formulation to reduce systemic toxicity.
Zosurabalpin
Zosurabalpin is a new macrocyclic peptide antibiotic that blocks LPS transport, killing CRAB and other highly resistant strains. Preclinical studies show strong activity in sepsis, lung, and thigh infection models with favorable safety. Human trials are still needed.
Zabofloxacin
Zabofloxacin is an oral fluoroquinolone effective for COPD exacerbations but weak against major nosocomial Gram-negatives like P. aeruginosa and A. baumannii. A phase III trial showed it was non-inferior to moxifloxacin. Approved in South Korea and some Middle-East/North Africa markets.
Imipenem/Cilastatin/Funobactam
This new β-lactam/β-lactamase inhibitor combination is active against serine carbapenemase-producing A. baumannii, P. aeruginosa, and Enterobacterales, including OXA-23/OXA-24 producers. A phase III HAP/VAP trial is comparing it with imipenem/relebactam to establish non-inferiority in mortality.
Limitations
Despite newer options, resistance is rapidly emerging. Mechanisms include overexpression of carbapenemase genes (e.g., blaKPC), metallo-β-lactamases, PBP mutations, and loss of iron-transport receptors like CirA, leading to resistance against drugs such as CAZ-AVI, ATM-AVI, and cefiderocol. Efflux pumps and modifying enzymes further contribute, especially in P. aeruginosa and CRAB. Because resistance varies across β-lactam/β-lactamase inhibitors, individual susceptibility testing is essential. Strong antimicrobial and diagnostic stewardship—guided by proper sampling, narrowing therapy, IV-to-oral switch, early discharge, and biomarker-based de-escalation—is critical to prevent resistance. Research is also supporting shorter antibiotic courses and inhaled/local therapies, though more evidence is needed.
Conclusion
A wide range of new antibiotics has emerged in the past decade to address rising antimicrobial resistance in respiratory infections. While these agents offer improved activity against MDR Gram-negative pathogens, careful antibiotic stewardship remains essential to prevent new resistance. Resistance patterns vary across β-lactam/β-lactamase inhibitors, so individual sensitivity testing is crucial. Combination therapies with older antibiotics are being explored, but current trials have limitations due to small, uneven patient groups. Larger, well-designed studies and real-world data are needed to define optimal use and improve patient outcomes.
Source: Papageorgiou, D.; Gavatha, M.; Efthymiou, D.; Polyzou, E.; Tsiakalos, A.; Akinosoglou, K. New Antibiotics for Lower Respiratory Tract Infections. Microbiol. Res. 2025, 16, 135. https://doi.org/10.3390/microbiolres16070135




Pingback: hello world
Pingback: diflucan over the counter spain
Pingback: viagra kopen
Pingback: metoprolol used for
Pingback: augmentin 875 mg
Pingback: nitrofurantoin 100mg capsules
Pingback: lasix generic
Pingback: lasix diuretic medication
Pingback: semaglutide vs tirzepatide side effects
Pingback: cialis 5mg reddit
Pingback: metronidazole flagyl oral side effects
Pingback: acyclovir where to buy