Introduction
Invasive fungal diseases (IFDs) present an increasing global burden in immunocompromised and other seriously ill populations. Early diagnosis encompassing accurate detection and identification of the causative agent and of antifungal resistance is critical for optimum patient outcomes. Panfungal PCR assays allow the rapid detection/identification of fungal species directly from clinical specimens with good specificity; sensitivity is also high when hyphae are seen in the specimen including in paraffin-embedded tissue.
Aspergillus PCR assays on blood fractions have good utility in the screening of high risk hematology patients with high negative predictive value (NPV) and positive predictive value (PPV) of 94 and 70%, respectively, when two positive PCR results are obtained.
Direct Detection of Fungi in Clinical Specimens
Panfungal PCR Assays
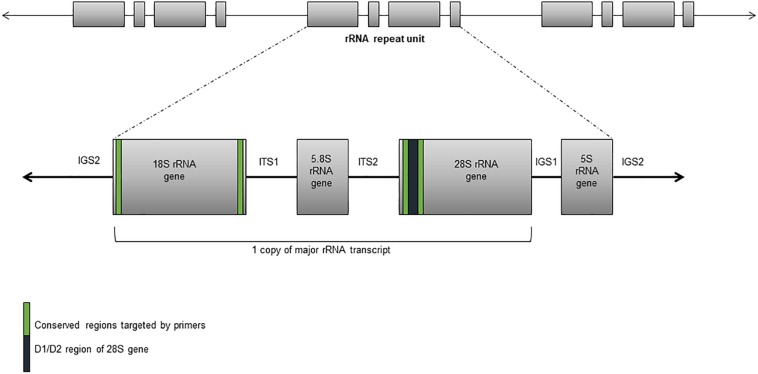
These assays detect “all” fungal DNA present in a clinical specimen through the use of universal fungal primers. The preferred target(s) are one or more regions of the rRNA gene cluster – the internal transcribed spacers 1 and 2 (ITS1 and ITS2) and the D1/D2 regions of the 28S rRNA gene (Figure 1).
Fig 1. Ribosomal RNA gene cluster comprising the 18S, 5.8S, and 28S rRNA subunit genes, and separated by the internal transcribed sequences and the intergenic spacers.
Together, these assays have successfully detected and identified fungi from diverse specimen types including fresh tissue, formalin fixed paraffin embedded (FFPE) tissue, cerebrospinal fluid (CSF), vitreous fluid, blood, and bronchoalveolar lavage fluid (BALF) with good accuracy and specificity though with varying sensitivity between specimen types.
Aspergillus PCR
Aspergillus PCR assays therefore have established utility in screening for early infection in high risk patients as well as being used in the diagnosis of established infection in real time. Greater clinical application may in the near future be realized by the growing number of commercial PCR assays to detect Aspergillus DNA in clinical specimens, providing standardized methodology, and quality control of the reagents (see Table 1).
Table 1. PCR-based assays for detection of Aspergillus spp. and CYP51A resistance mutations in A. fumigatus.
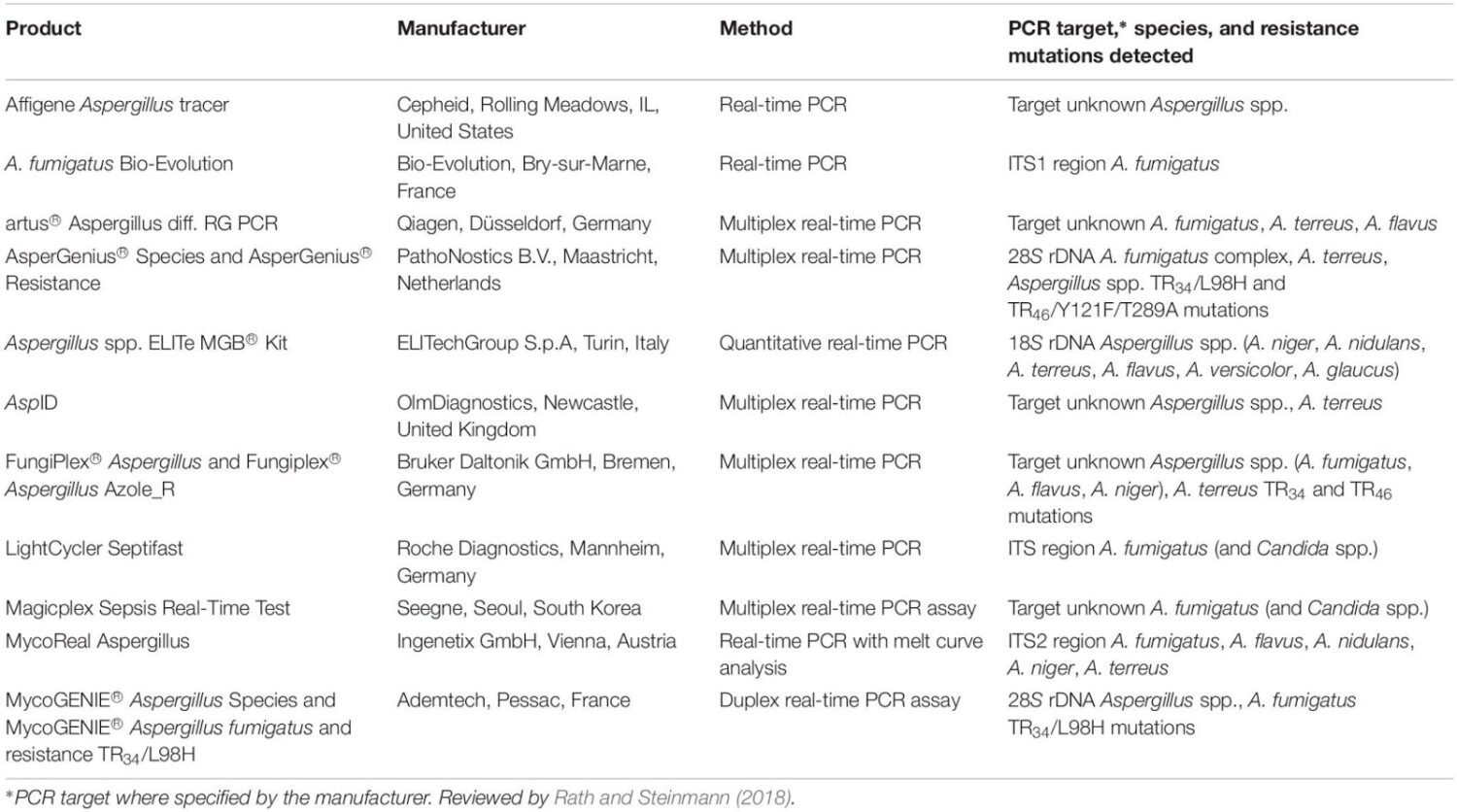
A recent review of commercial assays for the detection of Aspergillus spp. reported significantly lower sensitivities and specificities in serum specimens than respiratory specimens. Some assays simultaneously detect both Aspergillus DNA and the most prevalent CYP51A gene mutations responsible for azole resistance in A. fumigatus, differentiating wild type from resistant strains.
PCR for Invasive Candidiasis
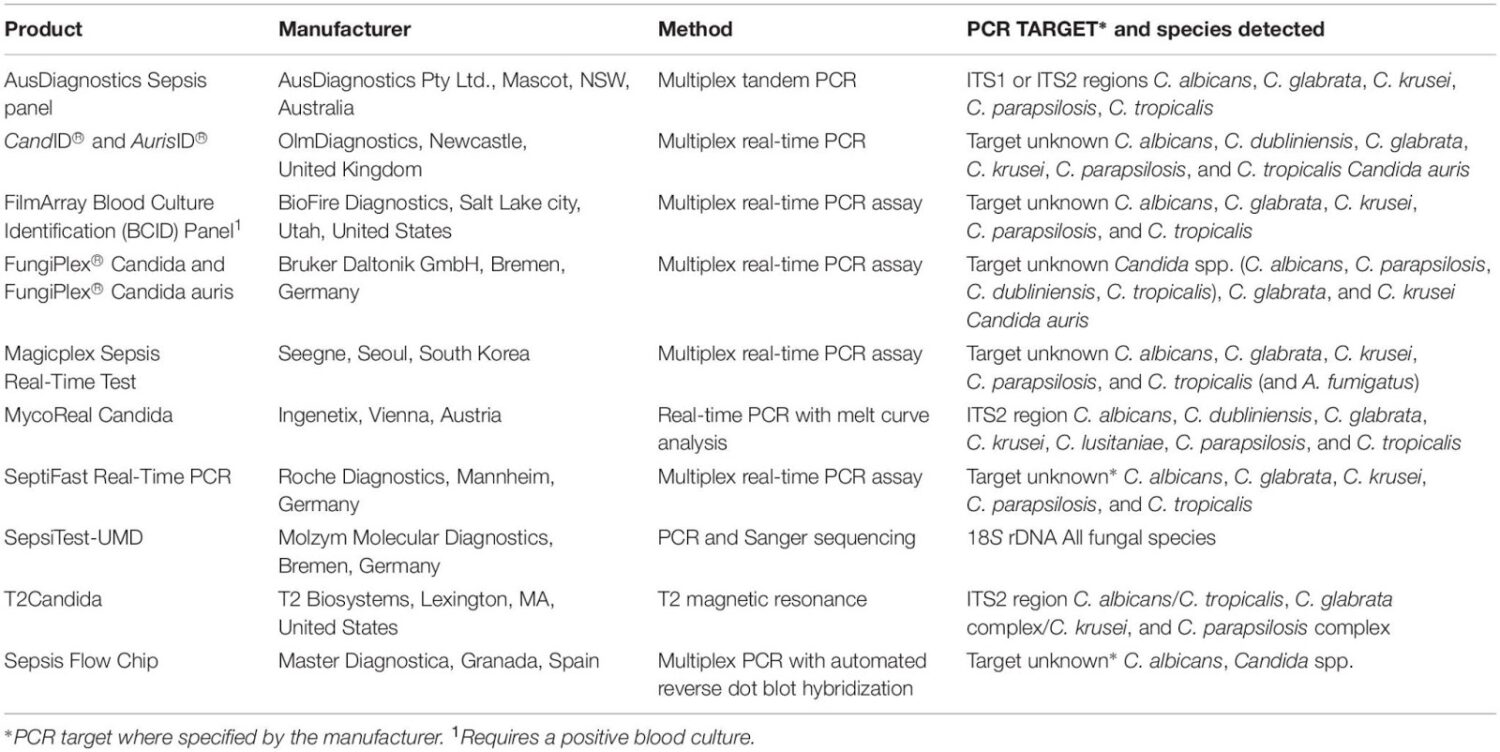
Generally, these assays target the five most common pathogenic Candida species which account for > 95%of IC. Many of these assays remain investigational as they have not been validated for diagnosing IC in multi-center studies, nor is there evidence that any one commercial test is superior.The choice of adopting an in house rather than commercial assay is dependent upon workflow and capacity in individual testing laboratories and cost, and results should be considered with caution. To date, the T2Candida panel and the T2Dx instrument is the only platform approved by the US Food and Drug Administration (FDA) to detect Candida spp. in WB without the need for prior blood culture or nucleic acid extraction steps.
The platform combines nuclear magnetic resonance and PCR technology for rapid (< 3 h), accurate, sensitive (1–3 CFU/mL), and specific detection of C.albicans/C. tropicalis, C. parapsilosis, and C.glabrata/C. Krusei. A number of commercial PCR-based diagnostic methods are now available for IC (Table 2).
Table 2. PCR-based assays for detection of Candida spp.
PCR Detection of Mucormycosis
Conventional and real-time PCR assays for the direct detection of minute amounts of Mucorales DNA from fresh or FFPE tissue, BALF, and serum have been reported with the majority targeting the ITS, 18S, or 28S rDNA with sensitivities in FFPE tissue of 56–91%. Real-time qPCR in serum and on average, can detect the pathogen 8 days. qPCR detection of Mucorales DNA in BALF is likewise an attractive approach to aid the diagnosis of pulmonary mucormycosis.
PCR assay for the detection of the clinically relevant Mucorales species enables direct detection of pan-Mucorales DNA, Rhizopus spp., Mucor spp., Lichtheimia spp., Cunninghamella spp., and Rhizomucor spp. in respiratory tract samples and fresh and FFPE tissue within 3.5 h. It is currently available for “research use only” purposes.
Pneumocystis jirovecii PCR
PCR-based assays for the diagnosis of Pneumocystis jirovecii pneumonia (PCP) undoubtedly are more sensitive than histological and microscopic identifica tion of asci and trophic forms in tissue, BALF, and induced sputum (IS) by conventional staining or by immunofluorescence microscopy.
A recent comparison shows an in-house qPCR method had sensitivity of 82.9% compared to 60.0% for the monoclonal immunofluorescence assay by the Mono FluoTM P. jirovecii IFA test kit with similar specificity (>99%).For pragmatic purposes, for more rapid TATs and because of steady loss of microscopy skills in the mycology laboratory, an increasing number of laboratories are turning to Pneumocystis PCR assays.
Molecular Detection of Antifungal Drug Resistance
Molecular Methods to Detect Azole Resistance in Candida Species
Azole resistance in Candida species can arise through: (i) point mutations in various Candida genes; (ii) upregulation of drug efflux pumps or transporters; (iii) overexpression of the drug target; and (iv) cellular stress response factors. Point mutations in the ERG11 gene, which encodes 14-alpha-demethylase, the target of the azoles, alters drug affinity and in C. albicans at least, also results in ERG11 overexpression via mutations in the transcription factor UPC2.
Mutations have been best described in three “regional hotspots” of ERG11 (amino acid positions 105–165, 266–287, and 405–488) and in the ERG3 gene (T330A and A351V), with again mutation studies best documented in C. albicans. These mutations can be reliably detected by allele-specific real-time molecular probes, DNA microarray technology, high-resolution melt curve analysis, and DNA sequence analysis.
Molecular Methods to Detect Azole Resistance in Aspergillus fumigatus
Molecular methods for detecting azole resistance in A. fumigatus is more straightforward than in Candida species as there is a strong association between specific mutations in the CYP51A geneF and the azole-resistant phenotype, rendering this locus a well-suited target for a molecular-based assay.
Amino acid substitutions in cyp51A are well described and in azole-resistant environmental isolates, mutations in the tandem base pair repeats of the promoter region of CYP51A coupled to amino acid substitutions in cyp51A leading to the following changes – TR34/L98H and TR46/Y121F/T289A. Other mechanisms of azole resistance include the overexpression of the CYP51A, and ABC and MFS genes as well as the gain of function mutation in the CCATT-binding transcription factor complex subunit hapE. However, the clinical relevance of these mechanisms is still uncertain.
Molecular Methods to Detect Echinocandin Resistance in Candida spp.
The application of molecular methods offers good potential to rapidly detect echinocandin resistance which hinges upon detection of specific mutations in the FKS genes of Candida. Echinocandins are not impacted by the actions of multidrug efflux transporters and the resistant phenotype is well understood to be a result of a number of amino acid substitutions in “hot spot” regions of the Fks subunits.
In Candida species, there are three FKS genes – FKS1, FKS2, and FKS3. Mutations in FKS1 alone will confer resistance in all species. In C. glabrata, mutations in FKS2 will also confer resistant phenotypes. DNA sequencing resolves all known FKS mutations within 3–4 hr. Alternatives include: (i) real-time PCR assays with or without probe detection which can distinguish WT strains from those with FKS mutations; (ii) microsphere-based assays using Luminex MagPix technology (Pham et al., 2014); and (iii) more sophisticated molecular beacon and melt curve assays.
A dual assay for C. glabrata FKS1 and FKS2 identified the predominant clinically relevant resistance-associated mutations in FKS1 and FKS2 within 3 hr. Finally, NGS is suited to detecting a large number of mutations in multiple FKS genes in clinical isolates of C. glabrata with high MICs to the echinocandins.
Concluding Remarks
Advances in molecular diagnostic technologies have undoubtedly improved the landscape for fungal diagnostics and identification. A range of tests for diagnosis and/or screening at risk patients for IFDs, with rapid turnaround times, and covering a broad range of IFDs have been researched.
Standardization of Aspergillus PCR has been included as a biomarker for IA in clinical trials. Molecular assays that directly detect Candida spp. from blood cultures or WB are used with other fungal biomarkers to inform the likelihood of infection. It is envisaged that species-specific assays will have a greater role to play in routine diagnostics, as will molecular assays that enable simultaneous detection of pathogens and their major resistance markers.
Finally, with high-level DNA barcoding, NGS technologies, and metagenomic approaches, the vision of a “one stop shop” for fungal biomarkers seems to be within reach in the foreseeable future. This wealth of data will require parallel studies to examine their clinical applications.
Source:
Sarah E. Kidd, Sharon C.-A. Chen, Wieland Meyer and Catriona L. Halliday. Front. Microbiol., 14 January 2020 | https://doi.org/10.3389/fmicb.2019.02903